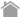- Japan(Japanese / English)
- Global
- MBL TOP
- MBL site search
How MHC Tetramer can detect T cells, and how to make it?
Reagent for direct detection of antigen-specific T cells
Class I and class II major histocompatibility complex (MHC) tetramer reagents allow rapid and simple detection of antigen-specific T cells. MHC tetramer technology is based on the ability of MHC-peptide complexes to recognize the antigen-specific T cells at a single cell level. This breakthrough technology enables researchers to precisely measure targeted T-cell responses in infectious diseases, cancer, and autoimmune diseases.
T-Select MHC Tetramers are available as ready-to-use reagents conjugated to a fluorochrome such as fluorescein isothiocyanate (FITC), phycoerythrin (PE), or allophycocyanin (APC). Whole blood, isolated peripheral blood mononuclear cells (PBMCs) , and expanded cell lines are appropriate sample types for tetramer analysis. MHC tetramers are used for the analyses of T cell immunity, as these reagents allow accurate enumeration and efficient immunomagnetic sorting of antigen-specific T cells, regardless of the T cell's functional capacity. MHC tetramers allow combined phenotypic and functional analyses of antigen-specific T cells using antibodies against surface and intracellular proteins. Monitoring of antigen-specific T-cell immune responses is thought to be the most important and relevant outcome of anti-tumor or anti-viral responses for the development of vaccines and therapies. These antigen-specific T cells can be detected using MBL T-Select MHC Tetramers.
Principle of Detection of Antigen-Specific T Cells by MHC Tetramers
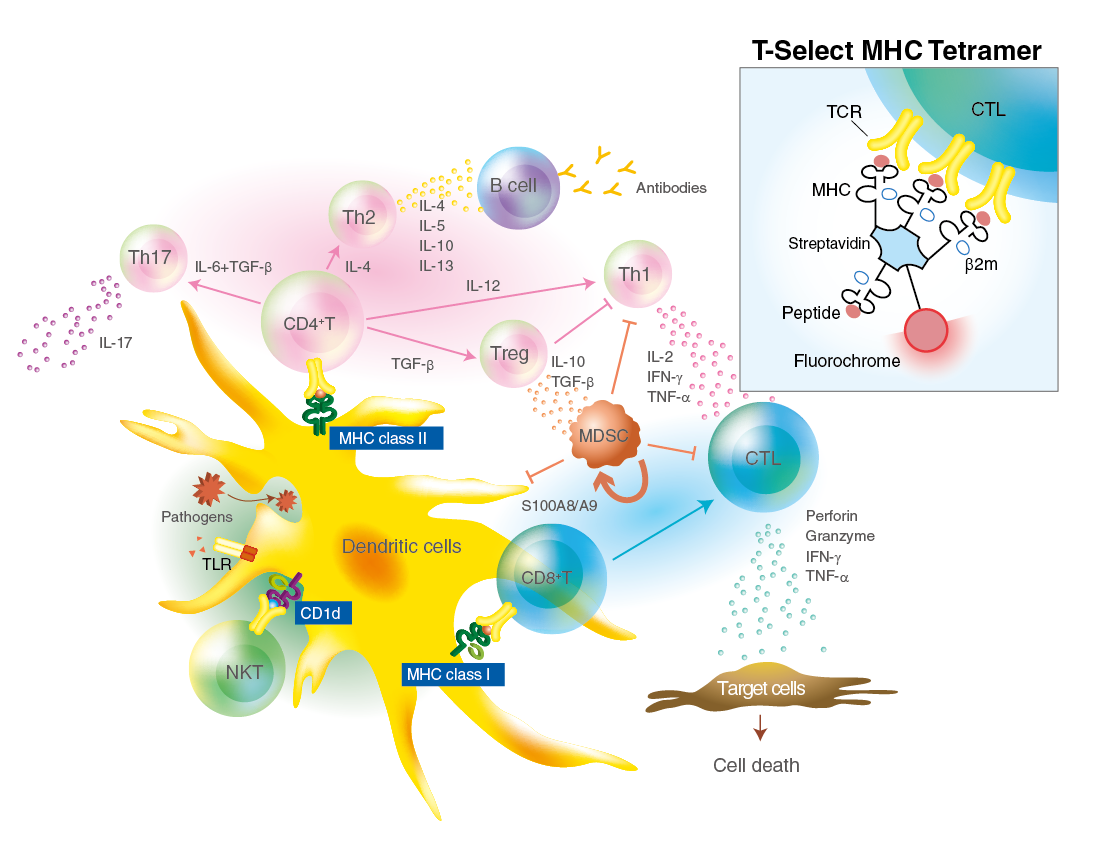
 Most T lymphocytes express a clonal and highly specific antigen receptor (TCR) on the cell surface. TCR (T cell receptor) binds to the MHC-peptide complex and recognizes the antigenic peptide presented in the context of MHC molecules. Such interaction initiates the generation of adaptive cellular immunity such as cell-mediated and humoral immunity. Because TCR has a low avidity and fast off-rates for MHC-peptide complexes, recombinant soluble monomeric MHC-peptide complexes have not been used to detect antigen-specific T cells. Altman et al.1) have introduced MHC-peptide tetrameric complexes (so-called MHC tetramers) for detection of antigen-specific T cells. MHC tetramers have increased avidity for their cognate TCRs and are successfully used to directly visualize antigen-specific T cells ex vivo by flow cytometry.
Most T lymphocytes express a clonal and highly specific antigen receptor (TCR) on the cell surface. TCR (T cell receptor) binds to the MHC-peptide complex and recognizes the antigenic peptide presented in the context of MHC molecules. Such interaction initiates the generation of adaptive cellular immunity such as cell-mediated and humoral immunity. Because TCR has a low avidity and fast off-rates for MHC-peptide complexes, recombinant soluble monomeric MHC-peptide complexes have not been used to detect antigen-specific T cells. Altman et al.1) have introduced MHC-peptide tetrameric complexes (so-called MHC tetramers) for detection of antigen-specific T cells. MHC tetramers have increased avidity for their cognate TCRs and are successfully used to directly visualize antigen-specific T cells ex vivo by flow cytometry.
Reference:
1) Altman JD et al. Science 274, 94-96 (1996)
Preparation of Class I MHC Tetramers
■ Steps for custom MHC class I tetramer
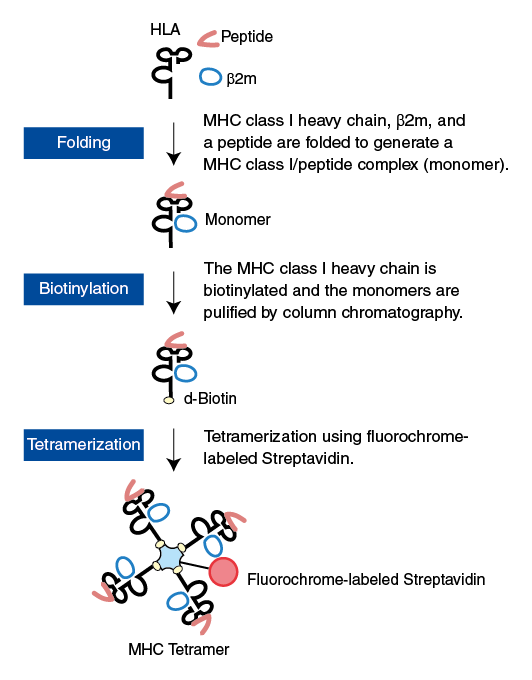
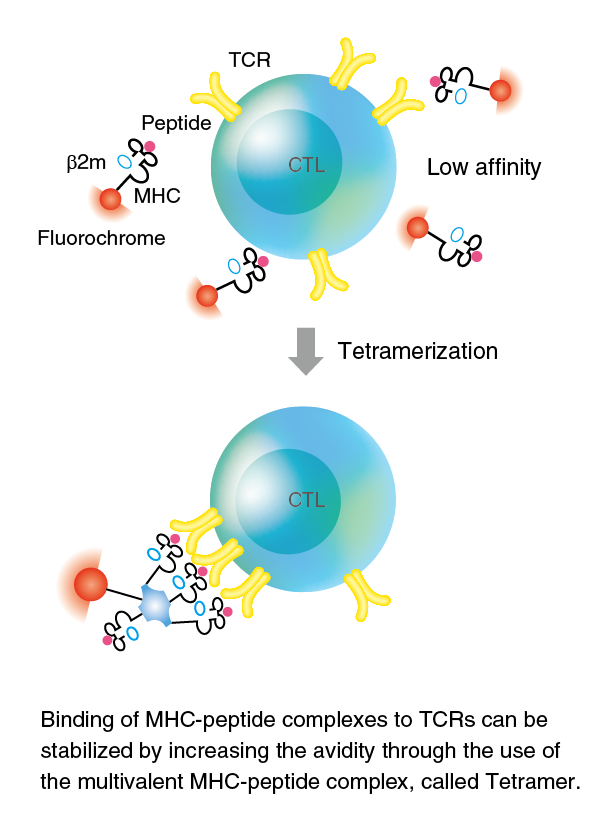
 MHC Tetramers are complexes of four MHC molecules, associated with a specific peptide and bound to a fluorochrome. The T cell surface CD8 enhances T cell antigen recognition by binding to HLA class I molecules. Therefore, we produced T-Select HLA class I Tetramers with one point mutation (Ala245Val) at the HLA α3 domain known to reduce the CD8-HLA interaction. These mutated tetramers showed a greatly diminished nonspecific binding but retained specific binding2). MHC Tetramers are generated essentially as described by Altman et al.1). The manufacture process is outlined in the figure on the right side.
MHC Tetramers are complexes of four MHC molecules, associated with a specific peptide and bound to a fluorochrome. The T cell surface CD8 enhances T cell antigen recognition by binding to HLA class I molecules. Therefore, we produced T-Select HLA class I Tetramers with one point mutation (Ala245Val) at the HLA α3 domain known to reduce the CD8-HLA interaction. These mutated tetramers showed a greatly diminished nonspecific binding but retained specific binding2). MHC Tetramers are generated essentially as described by Altman et al.1). The manufacture process is outlined in the figure on the right side.
Purified recombinant MHC (heavy chain) and human β2-microglobulin (β2m) are refolded in molar excess of the appropriate 8- to 10-mer peptide for several days. High-performance liquid chromatography (HPLC) has been extensively used to monitor the generation of monomeric MHC-peptide complexes (monomers). The refolded monomer is biotinylated with a single biotin by the BirA enzyme at the C-terminal end of the heavy chain. The biotinylated monomers are purified by streptavidin–agarose affinity column chromatography. Subsequently, purified monomers are linked by the addition of fluorochrome-labeled streptavidin.
Reference:
2) Bodinier M et al. Nat. Med. 6, 707-710 (2000)