- Japan(Japanese / English)
- Global
- MBL TOP
- MBL site search
Inflammation/Other marker
Lactoferrin ELISA Kit
 Lactoferrin is an iron-binding glycoprotein of the transferrin family that is expressed in most biologic fluids, such as the breast milk, particularly the colostrums, tears, sweat, saliva and other exocrine secretions of
mammals.
Lactoferrin is an iron-binding glycoprotein of the transferrin family that is expressed in most biologic fluids, such as the breast milk, particularly the colostrums, tears, sweat, saliva and other exocrine secretions of
mammals.
As a major component of mammals' innate immune system, lactoferrin has been shown to have numerous functions such as below.
1.“Antiviral effect”
By binding to the envelope of the Hepatitis C virus (HCV), lactoferrin
prevents infiltration of HCV to the target cells, and suppresses viral
replication.
2. "Antibacterial effect”
By chelating iron, lactoferrin suppresses the proliferation of iron
requiring bacteria.
3. ”Anti-inflammatory effect”
By strongly binding to endotoxin (LPS), lactoferrin inhibits the
binding of LPS to macrophages and suppresses the production of
inflammatory cytokines such as TNF-&alphs; and IL-6.
4. ”Immunoregulatory effect”
Lactoferrin stimulates cytotoxic activity of natural killer cells (NK
cells).
5. ”Antitumor effect”
Lactofferin induces the apoptosis of tumor cells, or prevent the
propagation of tumors by inhibiting angiogenesis.
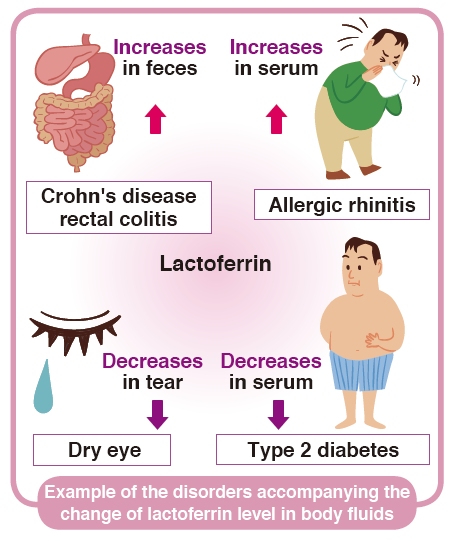
Focusing on anti-inflammatory effects, the lactoferrin in various biologic fluids are considered as markers of inflammation. It has been reported that fecal lactoferrin is useful as a sensitive and specific marker in identifying intestinal inflammation such as Crohn’s disease and chronic inflammatory bowel disease (IBD). Combination of several markers, such as S100A8/A9 complex, defensin, elastase, MPO and I-FABP may be useful for classifying IBD.
In addition, bovine milk prior to high temperature pasteurization (raw milk) contains large quantities of lactoferrin, and oral intake of lactoferrin has
been reported to cause several effects that maintain and promote health:
1. Proliferation of Lactobacillus biffidus
2. Improvement of anemia
3. Reduction of visceral fat
4. Prevention of arteriosclerosis by inhibiting the uptake of denatured LDL
Because of these effects, taking lactoferrin by milk products and supplements attracts public interest.
 CircuLex Bovine Lactoferrin ELISA Kit
CircuLex Bovine Lactoferrin ELISA Kit
14-3-3 Gamma ELISA Kit
 14-3-3 proteins are a family of conserved regulatory molecules expressed in all eukaryotic cells. There are seven genes, β, γ, ε, σ, ζ, τ and η, that encode 14-3-3s in most mammals. The family dynamically regulates the activity of target proteins in
various signaling pathways that control diverse physiological and pathological processes.
Either an abnormal state of 14-3-3 protein expression or dysregulation of 14-3-3/target protein interactions contributes to the development of many human diseases.
14-3-3 proteins are a family of conserved regulatory molecules expressed in all eukaryotic cells. There are seven genes, β, γ, ε, σ, ζ, τ and η, that encode 14-3-3s in most mammals. The family dynamically regulates the activity of target proteins in
various signaling pathways that control diverse physiological and pathological processes.
Either an abnormal state of 14-3-3 protein expression or dysregulation of 14-3-3/target protein interactions contributes to the development of many human diseases.
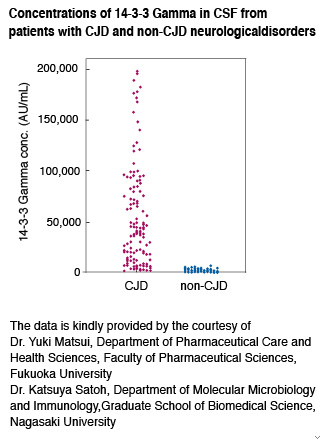
Clinical investigations have demonstrated a correlation between up-regulated 14-3-3 protein levels and poor survival of cancer patients. Several studies have also suggested that 14-3-3 isoforms are
differentially regulated in cancer and neurological syndromes. Especially, elevated amounts of the gamma isoform (14-3-3 Gamma) are found in the cerebrospinal fluid (CSF) of Creutzfeldt-Jakob disease (CJD) patients and could be as a specific marker of the disease as well as tau protein level.
 14-3-3 Gamma ELISA Kit
14-3-3 Gamma ELISA Kit
 14-3-3-related proteins
14-3-3-related proteins
DJ-1/PARK7 ELISA Kit Ver.2
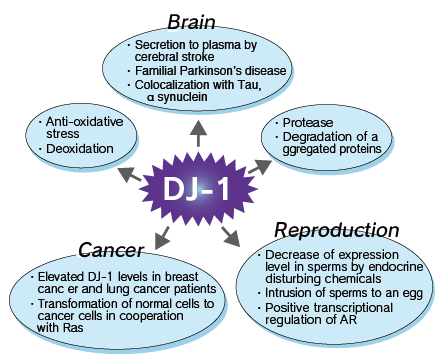 DJ-1 (PARK7/CAP1/RS) was originally cloned as a putative oncogene capable of transforming NIH-3T3
cells in cooperation with H-ras, a protein expressed in sperm, and a regulator of RNA-protein interactions. DJ-1 has also been isolated as a gene associated with autosomal early-onset Parkinson's disease (PD). Several lines of evidence suggest that DJ-1 plays a role in the oxidative stress response. In cultured mammalian ce11s, DJ-1 quenches reactive oxygen species and is converted into a variant with a more acidic isoelectric point. Therefore, DJ-1 protects against reactive oxygen species-induced cell death, and its suppression with small interfering RNA (siRNA) sensitizes cells to such insults. Mutations in DJ-1 that are associated with familial Parkinson's disease have been shown to decrease the anti-oxidative stress function.
DJ-1 (PARK7/CAP1/RS) was originally cloned as a putative oncogene capable of transforming NIH-3T3
cells in cooperation with H-ras, a protein expressed in sperm, and a regulator of RNA-protein interactions. DJ-1 has also been isolated as a gene associated with autosomal early-onset Parkinson's disease (PD). Several lines of evidence suggest that DJ-1 plays a role in the oxidative stress response. In cultured mammalian ce11s, DJ-1 quenches reactive oxygen species and is converted into a variant with a more acidic isoelectric point. Therefore, DJ-1 protects against reactive oxygen species-induced cell death, and its suppression with small interfering RNA (siRNA) sensitizes cells to such insults. Mutations in DJ-1 that are associated with familial Parkinson's disease have been shown to decrease the anti-oxidative stress function.
In addition, DJ-1 concentration is reported to be elevated within 3 hours after cerebral infarction, suggesting the possibility that DJ-1 could be a promising biomarker for early stage of cerebral infarction. Moreover, breast cancer patients have elevated levels of circulating DJ-1 and anti-DJ-1 autoantibodies compared to healthy and non-breast cancer patients.
ISG15 ELISA Kit
ISG15 (Interferon (IFN)-Stimulated Gene 15) is a ubiquitin-like protein containing two ubiquitin homology domains and becomes conjugated to a variety of proteins when cells are treated with type I interferon or lipopolysaccharide. Therefore this modification (so called, ISGylation) appears to be involved in type I interferon signal transduction. Unlike ubiquitylation, ISGylation does not drive protein degradation (regulated by K48-linked ubiquitin).
ISG15 can also be found extracellularly in an unconjugated form (free form). Human lymphocytes and monocytes were reported to release free ISG15 following treatment with IFN-β. More than 50 % of the total ISG15 could be detected in the culture medium after 24 hours following IFN stimulation. In addition, ISG15 is highly elevated and extensively conjugated to cellular proteins in many tumors and tumor cell lines. The increased levels of ISG15 in tumor cells were found to be associated with decreased levels of polyubiquitinated proteins.
 Human ISG15 ELISA Kit
Human ISG15 ELISA Kit
 Mouse ISG15 ELISA Kit
Mouse ISG15 ELISA Kit
 ISG15 proteins
ISG15 proteins
Fibulin-1 ELISA Kit
Fibulin-1 is the first described member of a seven-gene family of extracellular matrix (ECM) proteins
that have a common structural signature consisting of a series of repeated EGF-like domains followed by
a fibulin-type module at its carboxyl terminus. Fibulin-1 is a secreted glycoprotein that is an
intercellular component of a wide range of connective tissues. In blood vessel walls, fibulin-1 is a
component of elastic lamina and ECM fibers that surround smooth muscle cells and underlie the
endothelium. Although its actual function remains to be elucidated, fibulin-1 has been suggested to be
involved in cell adhesion, migration, proliferation, and malignant transformation through binding to
many ECM proteins, including laminin, fibrinogen, fibronectin, nidogen-1, endostatin, β-amyloid
precursor protein and proteoglycans, aggrecan, versican, receptors and growth factors. In
addition, fibulin-1 has been found to bind to the plasma protein fibrinogen and to incorporate into fibrin
clots formed in vitro and in vivo. Targeted inactivation of fibulin-1 gene in mice caused dilation and
ruptures in the endothelial lining of small blood vessels, indicating that fibulin-1 was important in
the stabilization of blood vessel walls.
Fibulin-1 is one of a few ECM proteins normally found in blood in high concentrations. It was
reported that plasma fibulin-1 concentrations increased in T2DM patients with prevalent cardiovascular
disease destined for vascular surgery as well as in asthmatics.
 CircuLex Human Fibulin-1 ELISA Kit
CircuLex Human Fibulin-1 ELISA Kit
High-Sensitivity CRP ELISA Kit
C-Reactive protein (CRP) is a pentameric acute phase reactant that is synthesized by the liver. Its production is controlled primarily by interleukin-6. The serum CRP concentration may increase by up to 1000-fold with infection, ischemia, trauma, surgery, and other acute inflammatory events. Thus CRP can be used as an extremely sensitive systemic marker of inflammation.
A growing number of studies suggest that CRP is an independent risk factor for atherosclerotic vascular disease. Plasma CRP concentrations in the highest quartile are associated, depending on the subject group, with 1.5- to 7-fold increases in relative risk of symptomatic atherosclerosis. The baseline plasma concentration of C-reactive protein predicts the risk of future myocardial infarction and stroke and is associated with a poor prognosis in unstable angina.
Plasma CRP levels are also strongly associated with obesity and obesity-related diseases, including insulin resistance, diabetes mellitus, and hyperlipidemia. Although a recent report indicated that the plasma CRP level decreased during weight reduction, the precise interaction of CRP with obesity has not been fully elucidated.
 High-Sensitivity CRP ELISA Kit
High-Sensitivity CRP ELISA Kit
Progranulin ELISA Kit
Progranulin (PGRN) also called granulin/epithelin, proepithelin, prostate cancer cell-derived growth
factor, acrogranin, or paragranulin, is a 593aa cysteine-rich protein that is typically secreted in a highly
glycosylated 88kDa form. PGRN is a growth factor involved in the regulation of multiple
processes including cell proliferation, tumorigenesis, wound healing, development and inflammation.
PGRN is widely expressed in epithelia, bone marrow, immune cells, solid organs, and the nervous
system both during development and in adulthood.
In 2006, mutations in PGRN were discovered to be a cause of frontotemporal lobar degeneration
(FTLD) with ubiquitinated TDP-43-positive inclusions. More than 70 mutations in
PGRN, almost all of which result in null alleles, have been identified in FTLD patients. A few causative
missense mutations also result in reduced levels of PGRN.
PGRN can be found in adipose tissue, epithelial tissue, gastrointestinal tract, reproductive organs, and
so forth. Previous studies have demonstrated that increased gene expression of PGRN stimulates
cancer cell division, invasion, and against anoikis, promoting tumor formation.
It has been shown that PGRN could restrain rheumatoid arthritis by binding directly to tumor necrosis
factor receptors (TNFR) and play an anti-inflammatory role in the processes. In addition, it was
reported that circulating PGRN levels are elevated in patients with type 2 diabetes. Moreover,
increased plasma PGRN levels are associated with impaired glucose tolerance rather than impaired
fasting glucose. Although type 2 diabetes is often accompanied by obesity, the respective role of
elevation of circulating PGRN levels in obesity and type 2 diabetes remains to be elucidated.
 CircuLex Human Progranulin ELISA Kit
CircuLex Human Progranulin ELISA Kit



 PCSK9 ELISA and PCSK9-LDLR in vitro Binding Assay Kits
PCSK9 ELISA and PCSK9-LDLR in vitro Binding Assay Kits